A report on myocarditis adverse events in the VAERS in association with COVID-19 injectable biological products

… 19 times the expected number of myocarditis cases in the vaccination volunteers
… a 5-fold increase in myocarditis rate was observed subsequent to dose 2 as opposed to dose 1 in 15-year-old males
… 67% of all cases occurred with BNT162b2
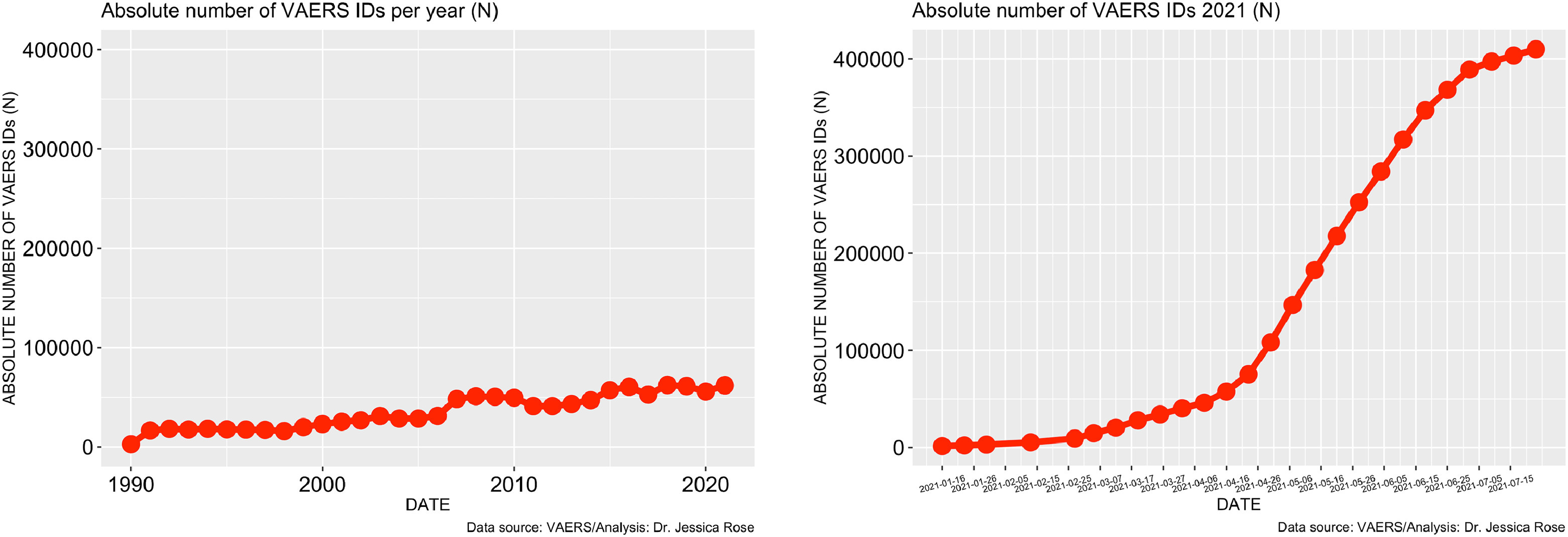
… far higher than last year for all vaccine products combined
… Because of the spontaneous reporting of events to VAERS, we can assume that the cases reported thus far are not rare, but rather, just the tip of the iceberg
… that children have a negligible risk for COVID-19 respiratory illness
… they are a high-risk group for myocarditis with vaccination
… to avoid children volunteering for injections with products that do not have proven safety or efficacy